The water used for filling sprayer tanks can affect herbicide performance, depending on water quality factors, such as pH, hardness, temperature, and soil particles. Credit: Doug Mayo, UF/IFAS
Pratap Devkota – Weed Science Specialist, UF/IFAS West Florida Research and Education Center, Jay, FL
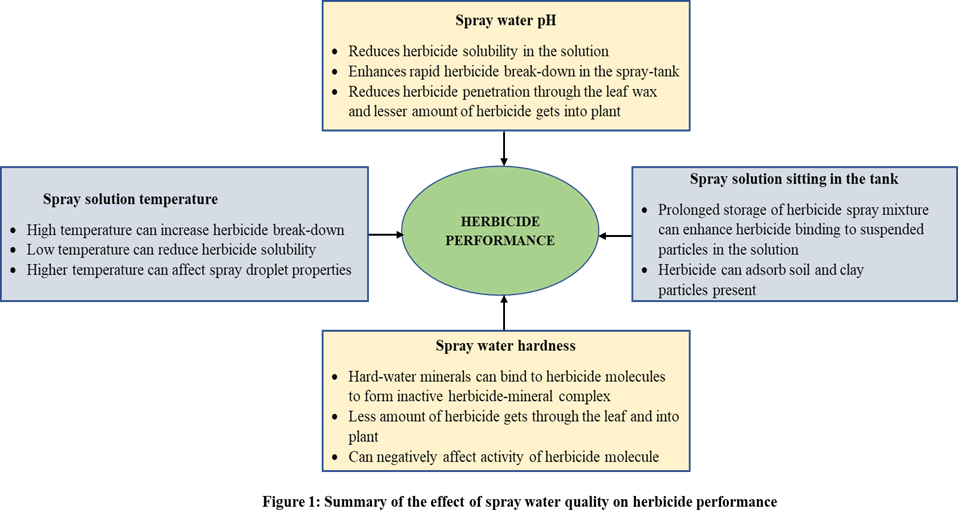
Water is the primary carrier solvent for herbicide application. Water used for herbicide application is obtained from various aboveground and underground sources. Depending upon the sources, the spray water differs in characteristics such as pH (acidity or alkalinity), hardness (the concentration of minerals), turbidity, and temperature. The inconsistencies in water quality factors could result in reduced herbicide performance through various mechanisms (Figure 1) and may need to be amended for optimum weed control.
–
Spray solution temperature and mixture storage duration effect on herbicide performance
Spray solution temperature can be variable depending on the ambient air or ground temperature. When herbicide solution is stored in a spray tank, the herbicide spray water tends to be at equilibrium with the prevailing ambient air temperature. A few studies have shown that spray solution temperature can adversely affect herbicide performance at the extremes. For example, spray solution temperature negatively influenced glufosinate, mesotrione, 2,4-D choline, and dicamba plus glyphosate efficacy at relatively low (41° F) or high (130° F) temperatures. But the efficacy was not affected at spray solution temperatures between 64° and 111° degrees F.
Unforeseen circumstances, such as unfavorable weather conditions, may prevent herbicide application immediately after mixing, thus prolonging the storage of a spray solution. The duration between herbicide spray mixture preparation and application can influence the performance of herbicides such as dicamba and 2,4-D. The longer the herbicide remains in the solution, the greater the possibility for herbicide molecules to breakdown or interact with constituents in the water. Prolonged storage of a herbicide mixture in the spray tank can also increase herbicide binding to the tank’s interior surface (made with polyethylene materials), which can result in herbicide breakdown over time and subsequent efficacy reduction on targeted plants. In general, if herbicide spray solution is applied within 8 to10 hours after mixing, there are no adverse effects on weed control. Lesser weed control could be observed if the herbicide solution must be stored overnight, depending on the herbicide used and the pH of the spray solution.
–
Spray water pH, hardness, and turbidity effect on herbicide performance
Spray water pH is one of the most critical water quality factors that can influence herbicide performance. Unfavorable water pH (acidic or alkaline pH) can adversely affect herbicide efficacy by affecting the solubility, enhancing breakdown of the herbicide molecules, which subsequently affects the herbicide’s ability to enter and move throughout the plant. In general, the efficacy of herbicide products containing glyphosate, glufosinate, clethodim, sethoxydim, bentazon, and 2,4-D are improved with an acidic water pH range. On the other hand, the efficacy of Group #2 herbicides (such as Cadre, Pursuit, Envoke, Sandea, Classic, etc.) are negatively impacted by acidic water pH, and need to be applied at alkaline pH for optimum efficacy. Similarly, the efficacy of Sharpen herbicide is improved when applied at alkaline water pH.
Water from different regions may consist of varying levels of minerals such as calcium, magnesium, iron, aluminum, zinc, manganese, etc., which contribute to water hardness levels. These cations can bind to the herbicide molecules and form a less soluble or inactive herbicide-salt complex, which does not readily get absorbed through the leaf wax. Eventually, this will result in a lesser amount of herbicide reaching the critical parts of the plant, reducing overall herbicidal activity. In some cases, the interaction of herbicide molecules and minerals can also increase the spray droplet size, which results in droplets bouncing off of the leaf. Glyphosate is the most widely studied herbicide with a hard water antagonism. Most of the research suggests a reduction in glyphosate efficacy on various weed species (velvetleaf, barnyardgrass, yellow nutsedge, broadleaf signalgrass, pitted morning glory, and Palmer amaranth) when applied with hard water. Likewise, other herbicides such as 2,4-D, Clarity, Enlist One, Enlist Duo, XtendiMax, Liberty, clethodim, and sethoxydim are found to be negatively affected by increased water hardness levels.
Inorganic (sand, silt, and clay) and organic matter (sediments) particles suspended in spray water can bind to herbicides and reduce performance. Water turbidity can reduce efficacy of various herbicides such as glyphosate, nicosulfuron, clethodim, sethoxydim, paraquat, diquat, etc. Herbicides that can bind tightly to the soil particles and have low carryover potential, such as glyphosate, paraquat, diquat are more prone to water turbidity effect.
–
Adjuvants for amending spray water factors and improving herbicide performance
Adjuvants are additives used for amending spray solutions and improving herbicide performance. Adjuvants also consist of spray buffers to adjust solution pH, water conditioners to amend hard water, drift retardants to reduce herbicide drift, and suspension aids to enhance the mixing of herbicide formulation. Adjuvants can improve herbicide performance by enhancing retention and absorption in plants. Ammonium sulfate (AMS) based water conditioning adjuvants are commonly used to overcome hard-water antagonism and improve herbicide ability to resist binding with cations in hard water. The sulfate anions of AMS can bind with mineral cations and prevent the herbicide-cation complex formation. Buffering agents are also used to modify or maintain spray solution pH and increase the solubility of herbicides in acidic or alkaline spray water. In general, pH buffer products and water conditioner adjuvants are recommended to overcome the negative effect of spray water pH or hardness. However, we do not have appropriate recommendations for specific herbicide products regarding the various spray water factors. Therefore, general guidelines could be implemented to address the effect of spray water factors for applying herbicides.
- Test water used for herbicide application for pH and hardness levels, simple kits are commercially available for quick tests.
- Do not use turbid or murky water for herbicide application.
- Read the herbicide label for available information on the water pH, hardness, and use of appropriate adjuvants, pH buffer, and tank-mix products.
- If water quality is a concern, consider using the highest labeled rate, lower spray volume, and bigger droplet nozzles for applying systemic herbicides. For contact herbicides, uniform coverage is most important, so you will need to spray at a higher spray volume.
- If water hardness is at a higher level (>180 ppm or mg/L), then you might need to consider tank-mixing appropriate water conditioners as permitted by the herbicide product label.
- While mixing, follow the order as: water conditioner adjuvants, pH buffer, dry herbicides, liquid herbicides, surfactants, and defoamer/drift retardants, etc.
- Check for any solute or precipitate formation after mixing herbicides. Make sure to check tank-mix compatibility of various products mixed.
- Do not delay application after mixing herbicide in the spray solution.
For assistance dealing with spray water concerns on your operation contact your county’s UF/IFAS Agricultural Extension Agent.