by Dana Stephens | Jul 7, 2025
PFAS—have you heard of them? Do you know what they are, or is it more of a term thrown around without much context?
PFAS are per- and polyfluoroalkyl substances. PFAS is a class of chemicals found in various industrial and consumer goods. For instance, you may find them in food packaging, textiles, cosmetics, and frequently in aqueous film-forming foams (AFFFs) used to extinguish fires. PFAS chemicals are known for repelling grease, water, and stains, making them widely used in various applications. These chemicals are stable and persistent, earning them the nickname “forever chemicals” because they do not readily biodegrade, or break down easily in the environment.
Numerous researchers suggested PFAS are abundant in aquatic systems and toxic to a range of aquatic organisms, with additional concerns of bioaccumulation of PFAS. PFAS accumulate in sediments and aquatic organisms, which pose health risks to wildlife and humans through the food chain. Research suggests linkages of PFAS to disruption of endocrine function, reproduction, and development in aquatic organisms. Research suggests similar linkages of PFAS to humans, like increased cancer risk, immune system suppression, endocrine and reproductive disruption, and child developmental concerns.
The United States Geological Survey (USGS) estimated at least 45% of the United States’ tap water has one or more PFAS chemicals (Smalling et al. 2023). At least one PFAS was identified in 60% of public wells and 20% of domestic wells supplying drinking water in the eastern United States (McMahon et al. 2022).
Have PFAS been found in Okaloosa County drinking water and surface waters?
Measured PFAS in Florida and Okaloosa County Drinking Waters
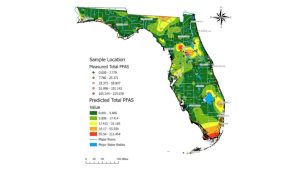
Figure 1. Map with measured total PFAS samples across a gradient of low concentrations (green dots) to medium concentrations (yellow dots) to higher concentrations (red dots). Shaded map colors are the predicted total PFAS using estimated values of PFAS concentrations from low (green) to high (red). Data, figure, and result interpolation from Sinkway et al. 2024.
A team of researchers completed a comprehensive statewide assessment of PFAS in Florida drinking water (Sinkway et al. 2024). The team collected 448 drinking water samples across all 67 Florida counties. The drinking water samples were analyzed for 31 PFAS, where 19 PFAS were found in at least one drinking water sample. The top five most frequently detected PFAS were 6:2 fluorotelomer sulfonate (6:2 FTS) (in 84% of the samples analyzed), Perfluorooctanoic acid (PFOA) (65%), linear perfluorooctane sulfonate (PFOS) (65%), branched PFOS (64%), and perfluorobutane sulfonic acid (PFBS).
A total of 107 taps had PFOA or PFOS concentrations above 4 ng/L (ppt), where the maximum total PFAS concentration in a tap was 219 ng/L (Click on link for higher resolution–Figure 1). The maximum contaminant level for PFOA and PFOS is 4 ng/L, legally enforced by the United States Environmental Protection Agency National Primary Drinking Water Regulation as of May 14, 2025 (USEPA, 2025). Overall, 8% of the drinking water samples analyzed exceeded 4 ng/L for PFOA and 16% for PFOS. The average total PFAS in city water was 15.6 ng/L, and in well water was 4.5 ng/L.

Table 1. Top average 12 Florida counties with highest and lowest total PFAS concentrations (ng/L). Data, figure, and result interpolation from Sinkway et al. 2024.
Okaloosa County had the 11th highest total PFAS (ng/L) concentration among the 67 Florida counties (Click on link for higher resolution–Table 1). Among the eight drinking water samples collected, the maximum PFAS concentration measured was 140 ng/L, and the lowest was 18 mg/L. Okaloosa County had one drinking water sample that exceeded the 4 ng/L standard for PFOA. There were no drinking water samples that exceeded 4 ng/L for PFOS.
Measured PFAS in Florida and Okaloosa County Surface Waters
A team of researchers completed a comprehensive statewide assessment of PFAS in Florida surface waters (Camacho et al. 2024). A network of citizen scientists collected 2,323 surface water samples across the 67 Florida counties. These surface water samples were analyzed for 50 PFAS, with 33 PFAS being detected in at least one surface water sample. The top five most frequently detected PFAS were perfluorooctanoic acid (PFOA) (94% of the samples), perfluorobutane sulfonic acid (PFBS) (65%), perfluorohexanoic acid (PFHxA) (61%), perfluorononanoic acid (PFNA) (54%), and perfluorooctane sulfonate (PFOS) (53%).
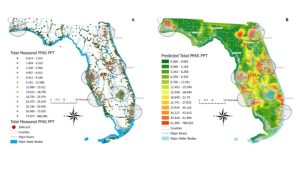
Figure 2. Map A contains all surface water sampled sites with detected PFAS, where the dots’ color represents the total PFAS concentration measured. Map B shows predicted PFAS levels based on measured total PFAS concentrations in surface water samples. Note that these values do not represent predicted PFAS concentrations on land. Data, figure, and result interpolation from Camacho et al. 2024.
There were 915 (39%) surface water samples with PFOA concentrations above 4 ng/L and 920 (40%) samples with PFOS above 4 ng/L (Click on link for higher resolution–Figure 2). All counties had at least one sample with PFOA, 96% had PFNA, 93% had PFBS, 91% had PFOS, and 82% of counties had PFHxA. The average PFAS detected among counties ranged from 2 ng/L of PFNA to 10 ng/L of PFOS. The maximum PFAS detected among counties ranged from 81 ng/L of PFOA to 1135 ng/L of PFOS. Figure 2
Okaloosa County ranked 27th among Florida counties due to 10 (20%) surface water samples with PFOA above 4 ng/L. Okaloosa County ranked 9th for the number of samples (38 total samples or 78% of the samples) above 4 ng/L for PFOS. A total of 49 surface water samples were collected in Okaloosa County. The average total PFAS concentration detected in a surface water sample was 31 ng/L, while the maximum total PFAS concentration detected in a sample was 185 ng/L.
Dr. Bowden, with the University of Florida’s College of Veterinary Medicine and Chemistry Department, led the PFAS research shared here. Dr. Bowden has extensive information on the Bowden Lab website (https://www.bowdenlaboratory.com/dr-bowden.html), including an interactive map of all the PFAS surface water samples collected in Florida. Select Okaloosa County under the filter section to see the surface water samples and learn more about the PFAS information for each sample collected in Okaloosa County (https://www.bowdenlaboratory.com/florida-surface-water.html).Figure 2 Table 1
What does this mean for Okaloosa County?
PFAS have been detected in drinking water and surface waters in Okaloosa County. Although not the highest concentrations or most frequent identified in Florida, there were drinking and surface water samples above USEPA’s 4 ng/L contaminant level standard. Understanding what PFAS are and joining in educational conversations about PFAS helps our community. Efforts that support continued sampling and extended monitoring also increase our understanding of PFAS concentrations in Okaloosa County’s drinking and surface waters. If you want to learn more about PFAS or join community scientists’ efforts to expand PFAS water monitoring, please contact Dana Stephens at the UF/IFAS Okaloosa County Extension Office.
References
Camacho, C.G., et al. 2024. Statewide surveillance and mapping of PFAS in Florida surface waters. American Chemical Society, 4: 434-4355. https://doi.org/10.1021/acsestwater
United States Environmental Protection Agency (USEPA). 2025. Per- and polyfluoroalkyl substances (PFAS) final PFAS national primary drinking water regulation. https://www.epa.gov/sdwa/and-polyfluoroalkyl-substances-pfas
McMahon, P.B., Tokranov, A.K., and Bexfield, L.M. 2022. Perfluoroalkyl and polyfluoroalkyl substances in groundwater used as a source of drinking water in the Eastern United States. Environmental Science and Technology 56(4): 2279-2288. https://doi.org/10.1021/acs.est.1c04795
Skinkway, T.D., et al. 2024. Crowdsourcing citizens for statewide mapping of per- and polyfluoroalkyl substances (PFAS) in Florida drinking water. Science of the Total Environment, 926: 1-9. https://doi.org/10.1016/j.scitotenv.2024.171932

by Dana Stephens | May 12, 2025
Spring is a time of change. Spring brings changes in our waters as well. Some of these changes are visible on top of the water and cause concern among water users and viewers. Let’s dispel some of these concerns associated with the spring season.
Sometimes, water users and viewers notice what appears to be oil floating on top of the water. Could this be oil? Potentially. Could this not be oil? Most likely. Plants perish, and decomposition occurs, typically during the spring and fall seasons of the year. Much of the decomposition that happens in spring is associated with the initial growth and development of plants. Bacteria living in the soils within and around the water break down the perished plants. These bacteria are decomposing the old plant material. The waste product produced from the bacteria’s decomposition of the old plant material is an oily substance. The oily sheen on the water is a waste product of bacteria. Frequently, the oil accumulates in portions of water where there is little to no water movement. As the decomposition process completes, the oily sheen should lessen over the next few days to weeks. This bacteria-produced oil from decomposition is a natural process.
Petroleum-based oil seen on water is not a natural process. Petroleum-based oil could enter water from various sources, such as but not limited to transportation spills, stormwater runoff, and improper disposal of products containing oil. Like the oily substances produced by bacteria during decomposition, petroleum-based oils will float on top of the water and accumulate where there is little to no water movement.
Here are some tips to identify the difference between oils in water:
|
Bacteria-produced Oil |
Petroleum-based Oil |
| Appearance |
Oily sheen on top of water with little to no difference in color throughout |
Oily sheen on top of water with differences in color throughout (may even appear like a rainbow) |
| Touch
(use a stick) |
When disturbed, the sheen breaks away easily with irregular patterns and does not reform. The oil will not adhere to the stick. |
When disturbed, the sheen swirls, elongates, and does reform. The oil may adhere to the stick. |
| Odor
(not always present) |
Strong organic, musty, or earthy smell. |
Volatile organic compounds (VOCs) smelling like gasoline or diesel fuel. |
Another sheen on our waters that is frequent during Florida’s springtime is pollen. Pine, tree, and weed pollen accumulate on top of water, especially in areas with little or no water movement. If the sheen on the water is yellow, orange, or sometimes white, this is most likely due to pollen. Think about how pollen shows on a car in Florida during spring…our waters can show the same to some extent.
Let’s give it a try! See if you can identify the sheens in water in each photo—answers at the bottom of the page.
Photo 1

Photo 2

Photo 3

Photo 4
Photo 5

Photo 6

Keep Scrolling For Answers!
.
.
.
.
.
.
PHOTO ANSWERS: Photo 1: Bacteria-produced oil sheen. Photo 2: Pollen sheen. Photo 3: Petroleum-based oil sheen. Photo 4: Pollen sheen. Photo 5: Petroleum-based oil sheen. Photo 6: Mixture of bacteria-produced oil and pollen sheen. Note all photos were obtained from Adobe Stock Photos.

by Dana Stephens | Apr 11, 2025
Okaloosa Waterwatch is an opportunity to explore water quality data in selected locations spanning the Okaloosa County portion of the Choctawhatchee Bay.
A site is selected each month, and available water quality data are summarized and explained. The purpose is to understand water quality and the condition of our waters. Thanks to the Choctawhatchee Basin Alliance (CBA), water quality data has been collected frequently (monthly) for many years.
Below are the February and March Okaloosa Waterwatch water quality summaries. February highlights the mid-bay bridge (Niceville) location, and March highlights the entrance to Rocky Bayou (Niceville) location.
Check out the Okalooas Waterwatch YouTube channel for a personalized audio review of the water quality summaries. The YouTube channel is Okaloosa Waterwatch (@OkaloosaWaterwatch) or navigate directly using this link: https://www.youtube.com/channel/UCGPVMsyMiTU5BT9xyrFhuYQ
Please contact Dana Stephens, UF/IFAS Okaloosa County Sea Grant Extension Agent, for more information or to learn more about water quality in Okaloosa County. Email is dlbigham@ufl.edu and office phone is 850-689-5850.
Okaloosa Waterwatch February 2025 (PDF Link)


Okaloosa Waterwatch March 2025 (PDF Link)

by Laura Tiu | Mar 7, 2025
 As spring approaches, I’ve been receiving more calls from local pond owners looking for advice on preparing their farm ponds for the season. Managing a pond in the Florida Panhandle can be tricky—especially when dealing with spring-fed ponds. While these ponds are often beautifully clear, their constant water turnover makes management a challenge.
As spring approaches, I’ve been receiving more calls from local pond owners looking for advice on preparing their farm ponds for the season. Managing a pond in the Florida Panhandle can be tricky—especially when dealing with spring-fed ponds. While these ponds are often beautifully clear, their constant water turnover makes management a challenge.
If you’re wondering how to get your pond ready for spring, here are some key considerations and resources to help guide you.
Start with a Water Quality Test
The first step in assessing your pond’s health is testing the water. I always recommend that pond owners bring a pint-sized water sample in a clean jar to their local Extension Office for analysis. Keep in mind that not all offices offer this service, and public testing options are limited. However, private labs and DIY testing kits are available—though they can be costly.
The most important parameters to check are pH, alkalinity, and hardness: pH should ideally range between 6 and 9 for a healthy fish population. Local ponds often hover around 6.5, making them slightly acidic.
Alkalinity and hardness measure the water’s ability to neutralize acids and buffer against sudden pH changes. For optimal pond health, alkalinity should be at least 20 mg/L, but many local ponds fall below this level.
Improving Pond Water Quality
If your pond’s water quality is less than ideal, there are two common ways to improve it: liming and fertilization.
Applying Agricultural Lime: Properly adding agricultural lime can raise alkalinity and stabilize pH levels. However, in high-flow ponds, lime tends to wash away quickly, making this method ineffective for ponds with constant discharge.
Fertilizing to Boost Productivity: Fertilization increases phytoplankton growth, which supports the pond’s entire food web, benefiting juvenile fish and invertebrates. Unfortunately, like lime, fertilizer is quickly washed out of high-flow ponds, making it ineffective in these cases.
Making the Best of Your Pond
If your pond has a continuous discharge due to spring flow, the best approach may be to embrace its natural clarity, even if it doesn’t support a thriving fish population. However, if your pond retains water without frequent outflow, you may be able to enhance its productivity with the right amendments.
For personalized guidance, contact your local UF/IFAS Extension Office. You can also start by reviewing this helpful fact sheet: Managing Florida Ponds for Fishing. By understanding your pond’s unique characteristics, you can make informed decisions to keep it healthy and enjoyable throughout the season.

by Andrea Albertin | Oct 18, 2024

Private well system in Florida. Image: UF/IFAS, C. Wofford
UF/IFAS Extension and Mississippi State University are offering a workshop about the fundamentals of private well and onsite wastewater systems (septic systems) on October 24, 2024 from 6-8:30 pm CST. Join us virtually or in-person at the UF/IFAS Walton County Extension Office for an informative session that will cover everything you need to know about these essential systems. Whether you’re a homeowner, realtor, or simply interested in the topic, this event is perfect for gaining valuable insights. Don’t miss out on this chance to expand your knowledge!
Training Objectives:
- Provide descriptions of the basic components of private wells.
- Explain basic private well maintenance and provide recommendations for water testing and treatment if issues are found.
- Provide descriptions of the basic components of onsite wastewater treatment systems.
- Explain how and why the different components of an onsite wastewater system exist and function.
- Explain how poorly functioning onsite systems can reduce the asset life of onsite systems and negatively impact water quality.
- Describe the impact inadequate treatment and discharge can have on nearby private wells and groundwater.
- Discuss private well and septic system best practices after flooding.
Date and Time: 10-24-2024 @ 06:00 PM (CDT) to 10-24-2024 @ 08:30 PM (CDT)
In-Person Location: University of Florida/IFAS Walton County, Walton County Extension Office; 732 N 9th St, Defuniak Springs, FL 32433
Trainers: Dr. Jason Barrett, Associate Extension Professor and the Associate Director, Mississippi Water Resources Research Institute at Mississippi State University, Dr. Laura Tiu, Marine Science Extension Agent, Walton County and Dr. Andrea Albertin, Regional Water Resources Extension Agent, University of Florida
Who Should Attend:
- Residents with private wells and onsite wastewater systems can benefit from the concepts covered in this presentation.
- Owners or managers of septic tanks, on-lot wastewater systems, and private wells.
- Municipalities and county officials overseeing local septic programs and decentralized wastewater systems.
- Soil and Water Conservation Districts (SWCDs) professionals.
- Professionals in water quality and wastewater management, including engineers, planners, and environmental consultants.
- Anyone seeking resources or guidance related to private wells, decentralized wastewater systems, and septic tank management.
Please contact the Walton County Extension Office (850) 892-8172 if you have any questions about the event.

by Rick O'Connor | Oct 4, 2024
One of the top concerns with residents in our communities is water quality. Surveys I have conducted with the public support this statement. One of the top concerns with water quality in the coastal areas is health advisories. These are issued when concentrations of selected fecal bacteria are too high.
In coastal areas, the selected bacteria are Enterococcus. It is important to note that Enterococcus bacteria are found in the digestive systems of birds and mammals. So, its presence does not automatically mean there is human waste in the water. However, there are pathogens associated with bird and mammal waste humans should be concerned about. And very high concentrations are most likely due to human waste. Science is working on additional methods to confirm high levels of Enterococcus are human. It is assumed that these will one day be used.
Enterococcus bacteria are used in coastal waters due to their ability to tolerate higher salinity. Some species, such as E. coli, that are used in freshwater systems die in saline ones. This suggests that the waste is not there, when in fact it is. It is also important to know that Enterococcus bacteria in themselves are not health concern for us. They are in our digestive tracts. But their presence in the water indicates that waste is present and there are pathogens in this waste that are of concern – hence the advisories issued.
In this series we will look at three methods we can use to help reduce human waste from entering our local waterways. Those are (1) maintaining your septic system, (2) converting your septic system to sewer, and (3) maintaining your sewer lines. We will begin with maintaining your septic system.
Septic systems have been used in rural and suburban communities where sewer systems have not been available for decades. Even today, the growth of new neighborhoods is outpacing the sewer infrastructure to support them. Many of these new communities are using septic systems. If properly placed and maintained, septic systems can work well. But many are not placed in good locations, and most are not maintained.
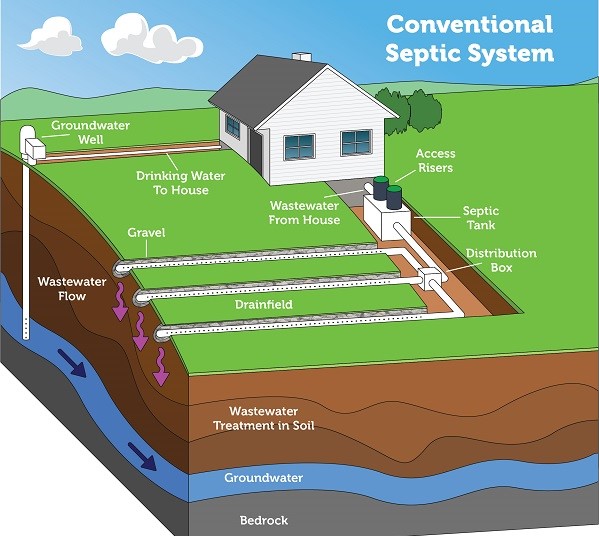
A conventional septic system is composed of a septic tank and a drainfield, where most of the wastewater treatment takes place. Image: US EPA
The system begins with the water leaving your home and entering a large tank made of concrete, fiberglass, or polyethylene buried in the yard. The average size of these tanks is 1000 gallons, but – depending on the number of bedrooms and bathrooms in the house, they could be larger. Here the sewage sits. Over time the solid waste will settle on the bottom (sludge) while the fats, oils, and grease float to the surface. The liquid layer in the middle (effluent) will flow from the tank into a series of smaller perforated drainpipes that slowly discharge into a drain field. A properly designed drain field will have a layer of sand that will allow draining of the effluent to occur.
Let’s talk about how to maintain this system.
- Do not overload the system with too much water. The tank is designed for a specific number of bedrooms/bathrooms. Overusing water can fill the tank and initiate leaking before the sewage has had time to settle. Watch your water use.
- Watch what you are flushing down the drain. Fats, oils, grease, and even milk will solidify and clog the lines. There are many products that describe themselves as “flushable”. They are, but they are not biodegradable. These two will cause clogs and backups in the system. Some harsh cleaning products can harm the bacteria within the tank who are breaking down the waste – these should be avoided. Also avoid using the garbage disposal. Septic systems were designed for water and sewage, not garbage and food. Compost your food waste instead.
- Periodically have your tank pumped and inspected. This is a step that many do not follow and can lead to leaking of untreated sewage into local waterways. It is recommended that you have your septic tank pumped and inspected once every 3-5 years. There are several businesses in your area who do this type of work. They can also provide advice on how to better maintain your specific system.
- Protect your drain field. Do not drive over this area of your lawn. Vehicles can compact the porous soil needed for efficient percolation and possible crack pipes. Planting trees and shrubs in this area can introduce roots into the lines, plant shallow root plants only. Design your landscape so that rainwater does not flow over the drain field during storms. The draining of your system is designed to be a slow process, allowing both physical and biological treatment of the waste to occur before reaching any water source. Rain and flooding conditions impede this from happening.
Developing a septic maintenance plan for your property can help reduce the number of health advisories your community will see. For more information contact your county health department or extension office.